Duchenne Muscular Dystrophy (Duchenne)
Duchenne muscular dystrophy (Duchenne) is a rare genetic disorder, caused by mutations in the gene responsible for making dystrophin, a protein involved in protecting muscle cell structure and function.
Duchenne primarily affects males, with approximately 1 in 3,500 to 1 in 5,000 boys affected worldwide. The absence of functional dystrophin protein in individuals with Duchenne results in cell damage during muscle contraction, leading to cell death, fibrosis, and inflammation in muscle tissues. Initial symptoms of Duchenne include muscle weakness that are often noticeable at an early age with diagnosis typically occurring by 5 years of age. Over time, individuals with Duchenne experience progressive muscle weakness and eventually lose the ability to walk. Respiratory and heart muscles are also affected, leading to difficulty breathing and the need for ventilator assistance, along with the development of cardiomyopathy. There is presently no cure for Duchenne.
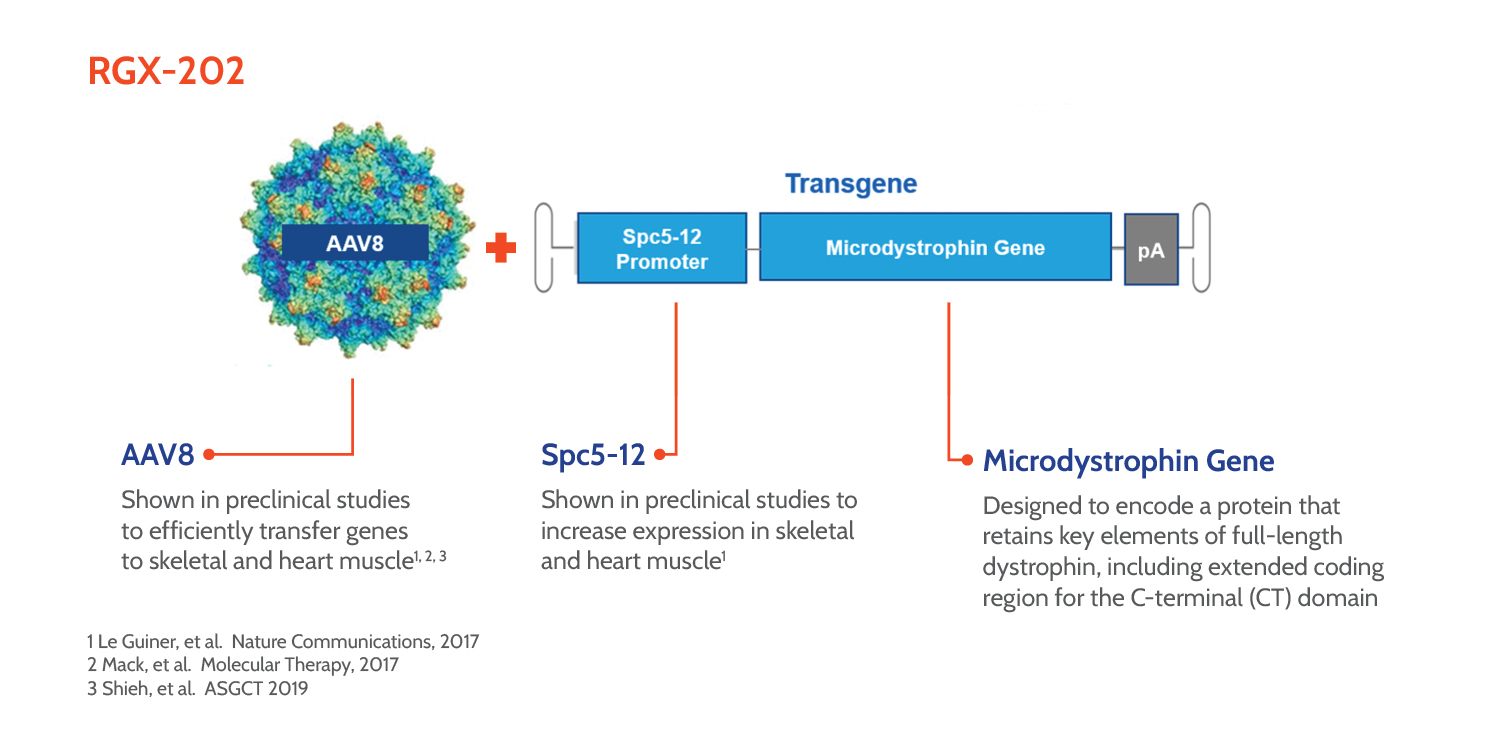
Our investigational gene therapy, RGX-202, uses REGENXBIO’s propriety NAV® AAV8 vector to deliver a transgene for a novel microdystrophin, a shortened and functional version of dystrophin protein. The RGX-202 novel microdystrophin includes functional elements of the C-Terminal (CT) domain for potential improved function. A well-characterized muscle-specific promoter (Spc5-12) has been shown in preclinical studies to selectively target expression of microdystrophin in skeletal and heart muscles. We believe that once the AAV8 vector delivers the microdystrophin transgene to muscle cells, they could produce the microdystrophin protein, protecting the cells from damage and, ultimately, preserving muscle function.
The pivotal Phase I/II/III AFFINITY DUCHENNE® trial of RGX-202 is active and recruiting patients aged 1 and above. Learn more about the trial: NCT05693142
The AFFINITY BEYOND® observational screening study to evaluate the prevalence of AAV8 antibodies in patients with Duchenne up to 12 years of age is also active and recruiting patients. Learn more about the trial: NCT05683379